 
Within a group (family) of elements, atoms form ions of a certain charge. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. This is also why ionic compounds have every high melting and boiling points.Ĭlick here to go back to the Science menu.\): Predicting Ionic Charges. The attractive forces are very strong in every direction that it takes a lot of energy to break them apart. This ions are regularly arranged in what is known as a lattice. In ionic compounds (compounds formed through ionic bonding), the strong attractive forces between the positive and negative ions result in the formation of a giant ionic structure. Describe the lattice structure of ionic compounds as a regular arrangement of alternating positive and negative ions, exemplified by the sodium chloride structure.Magnesium lost two electrons, so has a 2 charge and oxygen gained two electrons so has -2 charge. Now the configuration of magnesium is 2,8 and that of oxygen is 2,8. The quicker reaction is magnesium giving out the two electrons to oxygen. Oxygen needs to gain two electrons or give out 6 electrons. In order to be stable magnesium either needs to lose the 2 electrons or get 6 electrons. 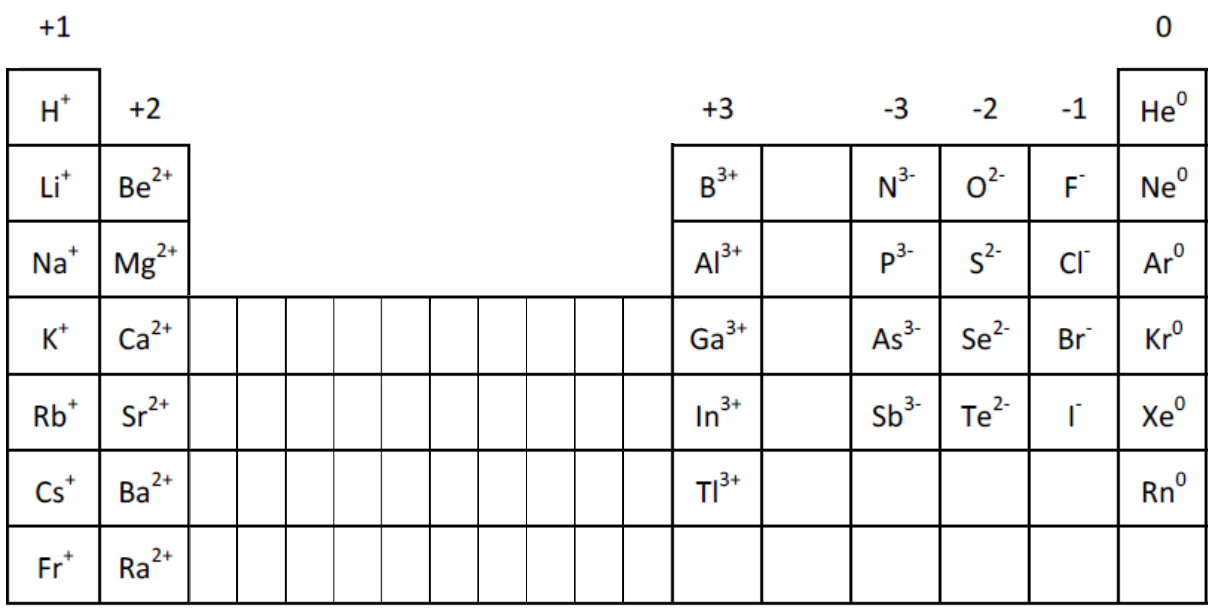
Magnesium has configuration 2,8,2 and oxygen has configuration 2,6. In the chemical bond created between metals and non-metals are always ionic because the metal (Sodium) becomes a positive ion and the non-metal (Chlorine) becomes a negative ion.Īnother example of ionic bonding bonding is the one between the metal Magnesium and the non-metal Oxygen. The above is an example of an ionic bond, where, oppositely charged ions are attracted. Explain the formation of ionic bonds between metallic and non-metallic elements.(The new compound formed is sodium chloride). Chlorine gained one electron, so it gained an extra negative charge, making it a negative (-1) Chlorine ion. Since sodium lost a negatively charged particle, it’s overall charge becomes positive, making it a positive ( 1) sodium ion. Both have full outer shells and so have become stable. This is reflected by decreasing half-lives for nuclei heavier than uranium. Now the configuration of Sodium will be 2,8 and the configuration of Chlorine will be 2,8,8. As the nuclear charge increases to large values, nuclei become more unstable. So sodium will have to lose it’s outer electron to chlorine. /PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png) 
In order to make them both stable, sodium needs to lose 1 electron and Chlorine needs to gain 1 electron. Let’s take the electronic configurations of metal ‘Na’ Sodium and the non-metal ‘Cl’ Chlorine.īoth elements are not stable. The elements in group 1 and 2 are metals and the elements in group 7 are non-metals. The block of elements in the yellow colour above are not considered part of any groups here). (Note that the columns in the periodic table are called groups.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
